Literature Reviews Made Simple
Whether you are a clinician, academic, or work in healthtech, systematic literature reviews can be an intensive task.
We empathise with you! Nevertheless, thoroughly reviewing the literature is vital when it comes to validating your tech. It will be the foundation of your technology’s intended patient population and target disease, benchmarking your tech’s performance, market access/competition strategy, and also clinical investigation design.
The basic definition of a systematic literature review
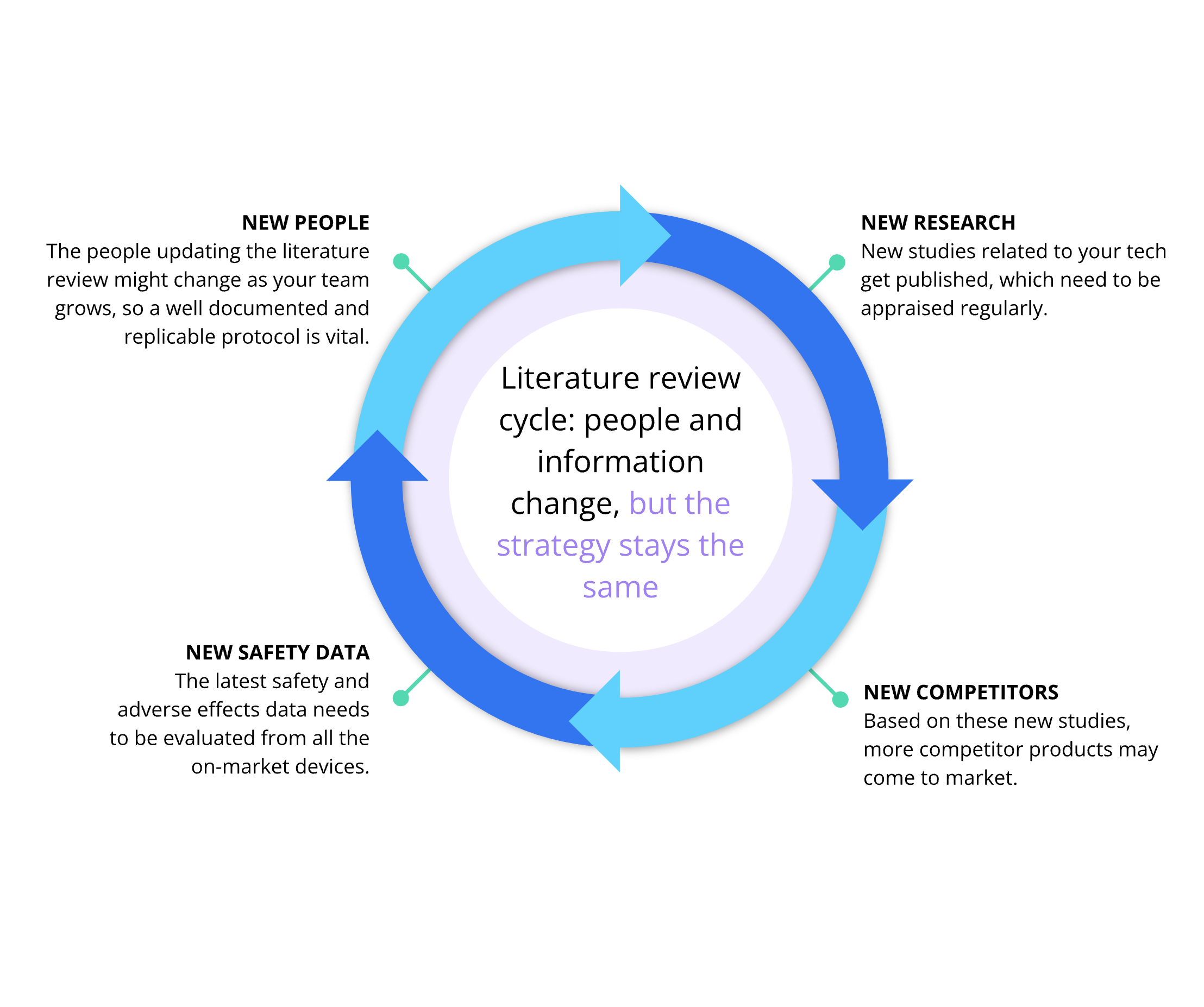
For those new to this, a literature review is a scoping exercise to find and critically evaluate the published scientific evidence as it relates to your product. A systematic literature review is one which follows a pre-specified and reproducible search protocol, and criteria to include or exclude studies for further evaluation. Taking a systematic approach is essential, because your review will need to be refreshed regularly to evaluate any new evidence. You need your methods to be consistent even as your team and product evolve.
Literature reviews for regulatory submission are different to academic systematic reviews
If you have a research background, it’s super important to understand the difference between the literature reviews you may have conducted in your academic field compared to your health-tech venture. In academia, systematic reviews and meta-analyses are often used to pool studies to answer a specific question, where the individual studies may have inconsistent outcomes. Sometimes they are also used to compile observational outcomes where these are poorly understood.
For regulatory submission, your literature review encompasses more than this. It should help you answer the following top-line questions as they relate to your product.
Does it work: is there a sound scientific basis for the technology or method that you are using for the disease in question? In other words, is there valid clinical association?
How well does it work (the technological gold standard): what are the reported performance metrics, and are there any known adverse effects you need to know about?
Is there anyone else doing this: who are your competitors/equivalent devices, and what is their published performance? Are there any reported risks or adverse effects of the deployed solutions?
What’s the current clinical gold standard: how is this problem currently being solved by clinicians, and what are the national or professional recommendations that need to be considered when deploying your solution?
What are the gaps in our knowledge: is there any scenario in which you intend to use your technology where there isn’t good evidence to support it?
When should you do your literature review?
Ideally, doing a literature review should be the next step after defining your product’s intended use. Since the outcome of your review will inform the studies you design, it needs to be done early in the product life cycle so that you can identify and plan to address any gaps in the evidence.
Once your product is deployed, it’s important to refresh your review at regular intervals, based on how quickly new information (including adverse events data) tends to accumulate in your field. As a result, it’s important to have a well-documented and systematic protocol for conducting your review. The PRISMA guidelines represent the scientific gold standard for systematic reviews. Within this framework, it’s also important to appraise the quality of individual studies (e.g. using the GRADE methodology). This makes sure that no evidence is missed, and that the results can be easily replicated through each update cycle.
Why do you need a high quality, systematic, literature review?
The simple answer is that you need it because the regulators require it, but we really encourage you to treat it as more than just a ‘tick box’ exercise. At Hardian we often see that a well conducted literature review helps companies understand how they may need to modify their product to maximise their chances of success, as well as identify what remains to be proven in future investigations. Most importantly, it’s just part of serving patients in a safer and more effective way.
Hardian Health is clinical digital consultancy focussed on leveraging technology into healthcare markets through clinical strategy, scientific validation, regulation, health economics and intellectual property.